 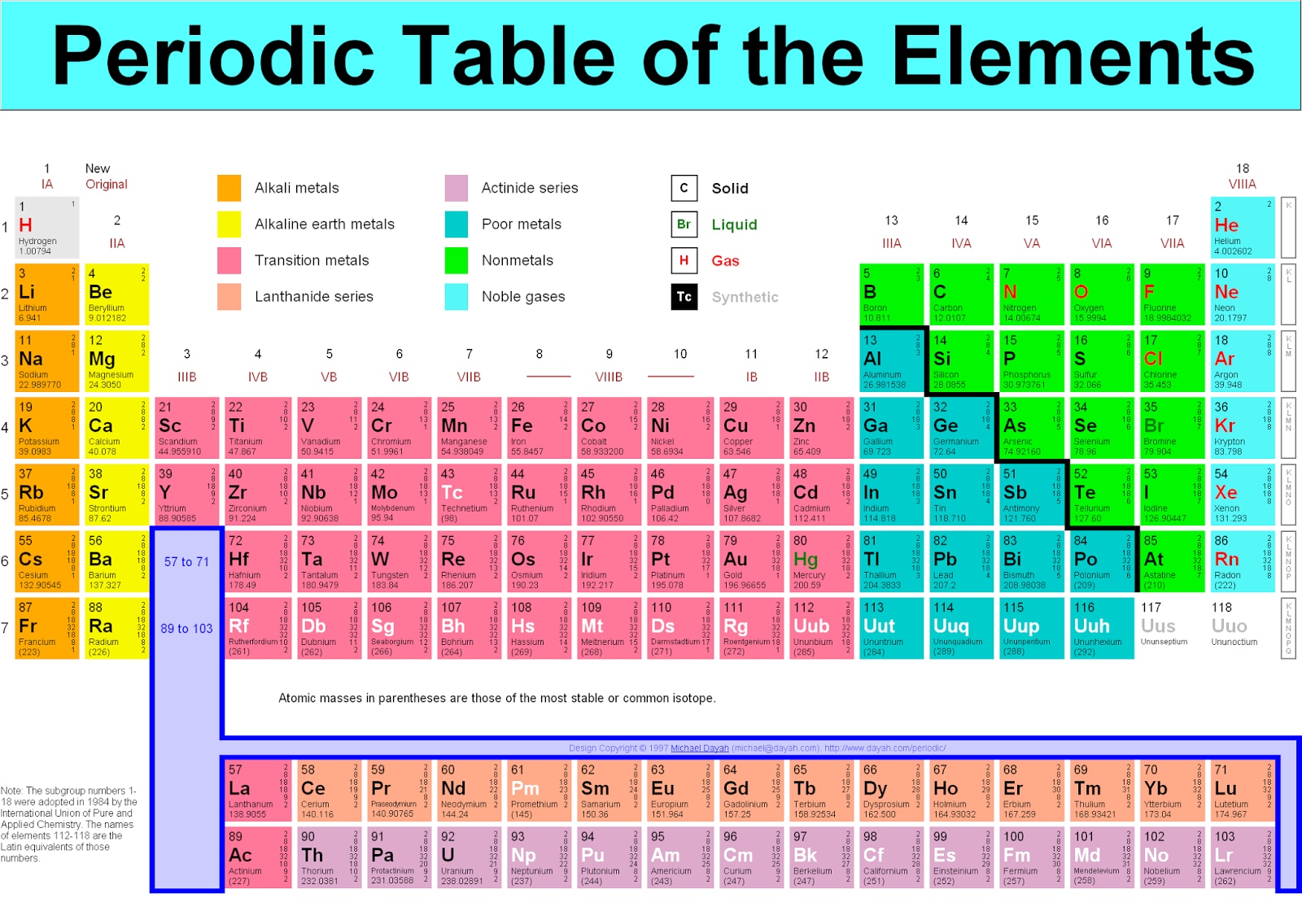
Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#). The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. On the other hand, the molar mass is the mass of the one mole of particles ( atoms or molecules) expressed in grams per mole (g/mol). 
One a.m.u is the mass of an element relative to the 1/12 mass of a carbon atom. Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula Atomic mass, also known as atomic weight is the mass of an individual atom expressed in atomic mass units (a.m.u). The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Remember thats the number written under the element symbol and element name. So in most cases, to find the molar mass of an element, you just need to look at its atomic mass (atomic weight) on the periodic table. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: The molar mass of an element is the mass in grams of one mole (6.02 x 10 23 particles) of the element. Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: A row on the periodic table is called a period. Heres a nice Periodic Table and more pdf for you to use for this class. I prefer to use tables with whole number atomic masses, but that is just personal preference. The periodic table is your ultimate conversion chart for converting any substance into another substance and doing so with exact proper amounts (masses and moles). Find a periodic table and look at the atomic mass of each atom in the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
